
This is why seawater at Antarctica is very salty. Under these conditions, the exact density of water is 999.972 kg/m 3. The salt that is rejected forms brine beneath the ice and becomes more and more salty until it becomes so dense that it sinks, displaces less dense seawater that moves to the surface. The maximum density of water is reached at a temperature of 3.84.2☌. 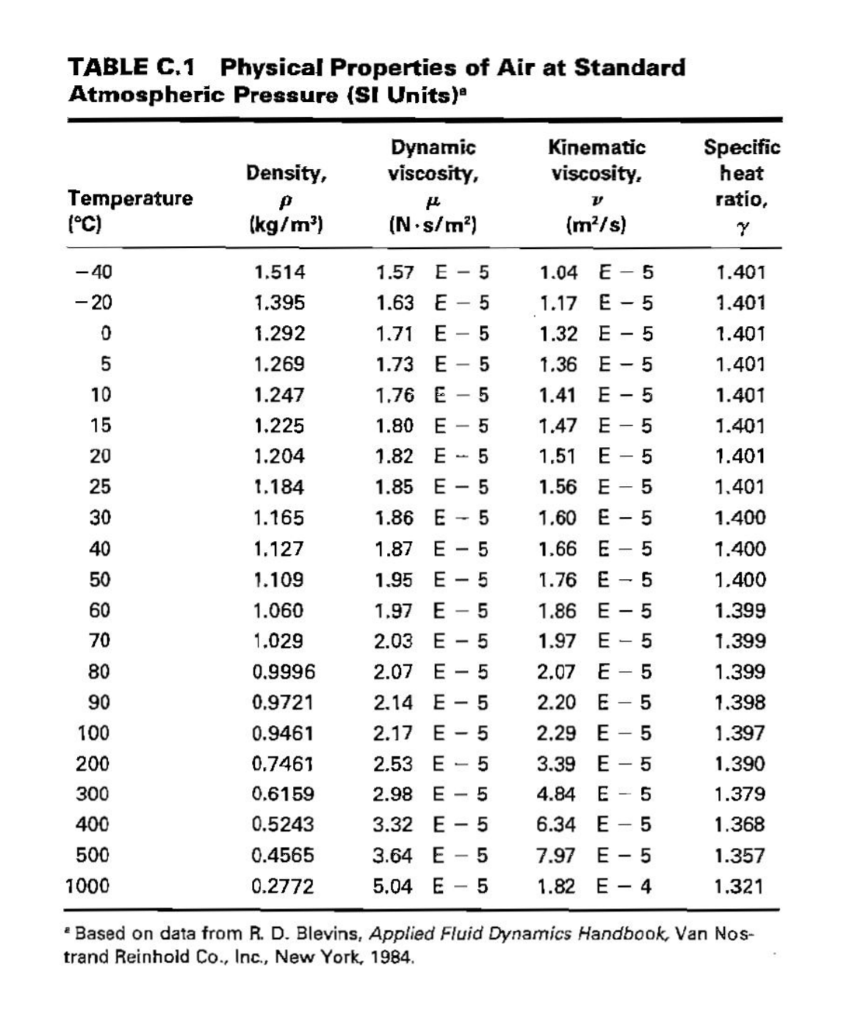
As ice forms in the sea, the salt cannot form part of the ice crystal so the ice is almost pure water. Density of water and ice Density of ice and water as a function of temperature.The salinity of seawater is about 3.5% and it freezes at about -1.9☌.The addition of salt to water makes a solution that is denser than fresh water – it freezes at a lower temperature.The hydrogen of the water molecule is attracted to chlorine ions and the oxygen to the sodium ions. This allows the sodium and chlorine ions to be pulled apart by the water molecules. Salt dissolves in water because the attraction between the water molecules and the sodium ions or chlorine ions is stronger than the attraction between the sodium ions and chloride ions in the lattice. Density (g/cm 3) State of Matter hydrogen : 0.00009: gas: helium (at STP) 0.000178: gas: carbon monoxide (at STP) 0.00125: gas: nitrogen (at STP) 0. 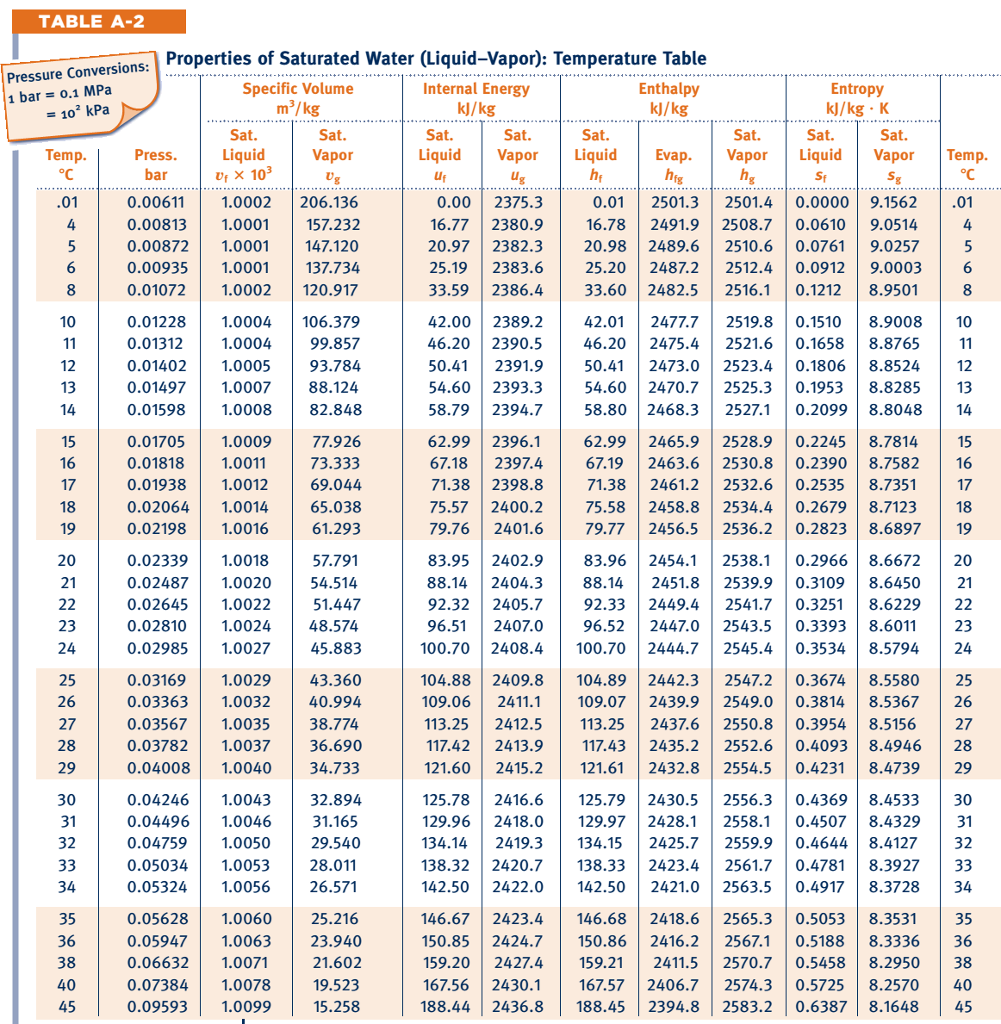
For every sodium ion you will find one chlorine ion (1:1 ratio). Salt is made up of many sodium and chlorine ions stacked together in a lattice.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
